Finding the density of solids is much easier than you think. First lets looks what the density is?
Density is defined as mass per unit volume:
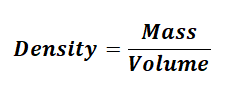
So in order to find the density of solids, all we need is to measure the mass and volume of the solid sample and then divide mass by volume.
Measuring the mass
To measure the mass, we need to the sample on weighing scale and take the reading.
Measuring the volume
In 3rd century BC, Archimedes, a famous Greek mathematician was exactly pondering on the same problem that is how to measure the volume of the crown as the King assigned him the task of determining whether his new crown is made up of pure gold (as the jeweler claimed) or not.
Archimedes plan was to find out the density of gold crown and compare it with the density of pure gold to find out whether the jeweler claimed is correct. He knows how to measure the mass of the crown by weighing in however he could not figure out how to measure the volume of the crown as it has a complex shape.
One day Archimedes was about to take a bath and as he stepped into the full tub, he saw water overflowed. At this point, he immediately realized that the volume of the body that was submerged was equal to the volume of the water. He got so excited that he ran home naked through the streets yelling “Eureka, Eureka!” to solve his remaining puzzle of finding the density of the King’s crown.
Measuring Density
In order to find density of solid, we will be requiring following things:
- Solid sample whose density is to be measured.
- A weighing scale
- A beaker with partially filled water.

Step-1 Measure the mass of the solid sample by weighing it on a weighing scale.

As shown on the weighing scale, the mass of the ball is 32g.
Step-2 Now take a beaker and partially fill it with water.

As shown from the above picture, the beaker is filled with 200 ml of water.
Step-3 Place the solid sample into the beaker of water. Now you will see the level of water will be raised.

Step-4 Take the reading of this new volume from the beaker. As shown in the picture below, the level of water has now be raised at around 212ml.

Step-5 In order to measure the volume of the solid sample, all you have to do is to subtract the volume of water without solid sample from volume of solid sample immersed in water.
Volume of Solid Sample = Volume of water with solid sample – Volume of water without solid sample
As we now have mass and volume of the solid sample, density of the solid sample can be found out by dividing the mass by volume.
We can calculate the density of Quartz ball as follows:
Mass of the ball (m) = 32 g
Volume of the ball (V) = 12 ml = 12 cm3
Since,
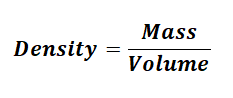
So Density of Solid Sample = 32g / 12 cm3 = 2.67 g/cm3
This density is closely related to Quartz crystal which is understandable because the solid sample used is made up of Quartz crystal.
Calculate Mass, Volume or Density using this simple Calculator
